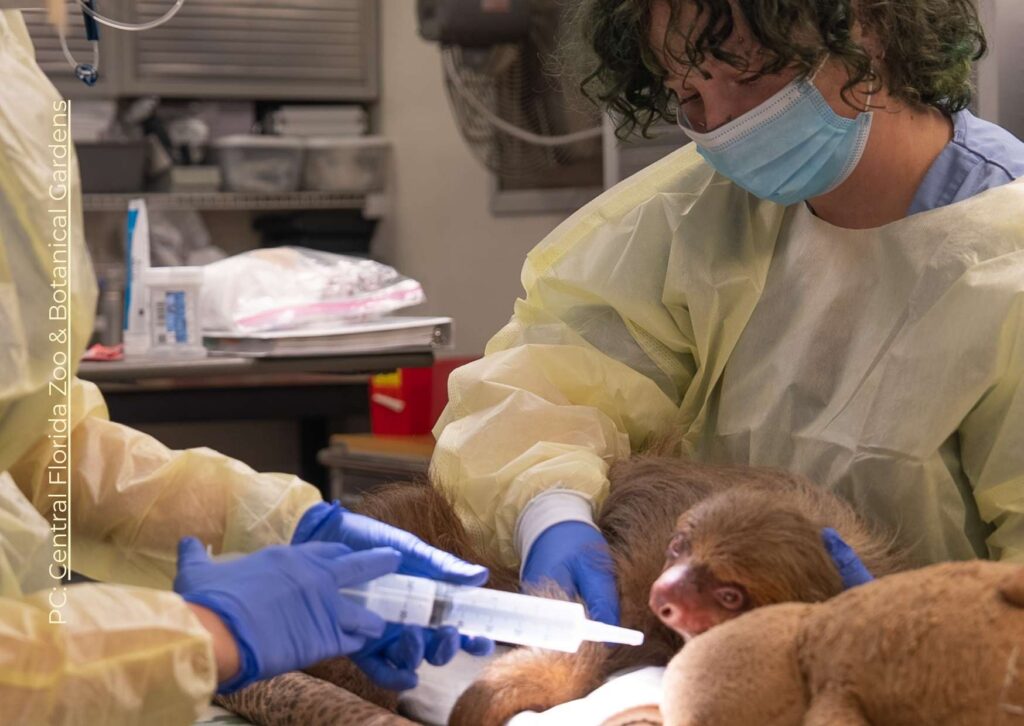
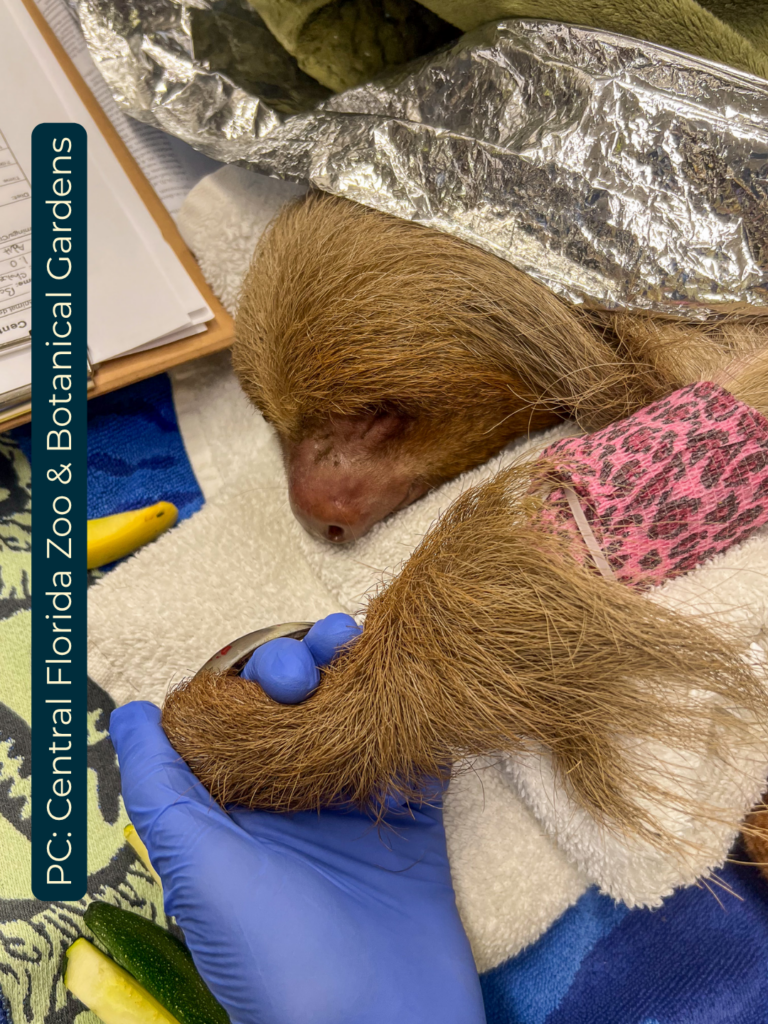
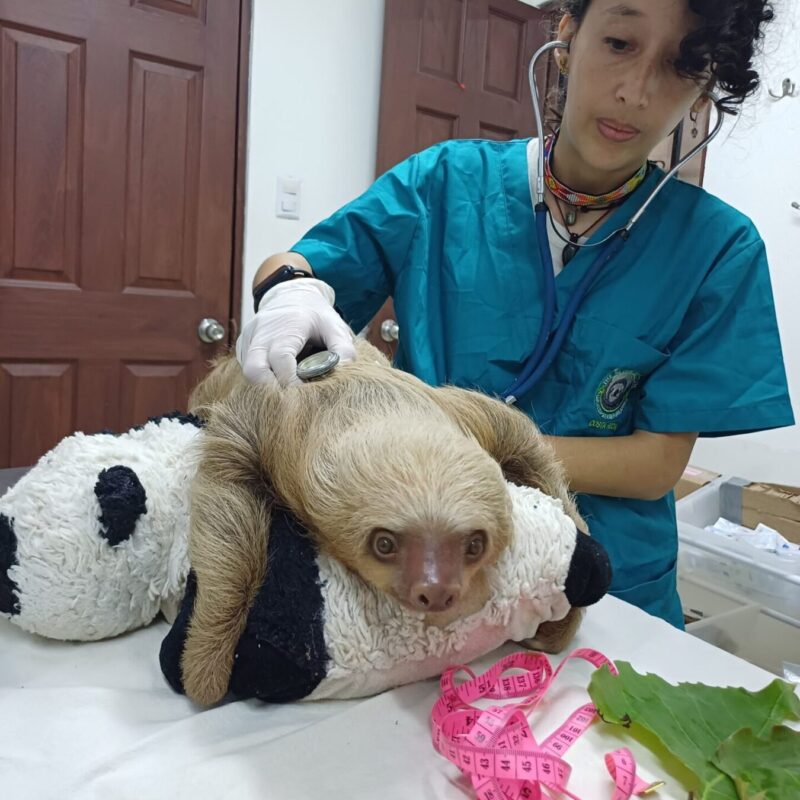
These records document a mass mortality event among a group of two-fingered sloths following their transport from Peru to Florida in 2025 and 2026. Veterinary clinical notes and necropsy reports from the Bronson Animal Disease Diagnostic Laboratory describe a wide range of fatal conditions, including severe diarrhea, pneumonia, and organ failure. Pathologists identified several contributing factors, most notably a novel gammaherpesvirus likely triggered by the extreme stress and cold temperatures of travel.
Between August 2025 and April 2026, the Sloth World facility in Orlando, Florida, experienced a significant mass mortality event involving two-fingered sloths (Choloepus spp.). The initial cluster followed a transport from Peru on August 13, 2025, where sloths were subjected to environmental stressors, including an overnight stop in Lima with temperatures averaging 60°F (15.56° C).
The creator of the reports has the theory that the primary driver of mortality has been identified as a novel two-fingered sloth gammaherpesvirus, which the necropsy report attempts to attribute to severe interstitial pneumonia and a systemic illness. Diagnostic investigations suggest that the stress of capture and transportation likely induced systemic stress that caused the death of the individuals, although not everything can be attributed to the viral disease.
Secondary contributing factors include:
- Systemic Pathology: Consistent findings across necropsies included severe hematopoietic depletion in bone marrow, gastric dilation, and lymphoreticular depletion.
- Environmental/Transport Stress: Potential capture/transport myopathy and immunosuppression from cold exposure.
Clinical History and Transport Stress
On the evening of August 13, 2025, a shipment of 10 sloths arrived from Peru. The transport included an overnight stay in Lima in 60°F temperatures. Upon arrival, several sloths exhibited severe diarrhea and fecal staining.
Initial Clinical Presentation
The medical logs for the “Crate” sloths (Flash, Belt, Selma, Slakoth, Leif, Baloo, Mossy, Ceviche, Poncho, and Sid) show a rapid progression of symptoms:
- Gastrointestinal: Severe watery diarrhea, gastric bloat, and anorexia.
- Neurological/Systemic: Lethargy, dilated pupils with no pupillary light reflex, and weakness.
- Thermoregulation: Several sloths (Flash, Slakoth, Leif) exhibited dangerously low body temperatures (84°F–88°F) despite being provided with 90°F heat support.
Mortality Timeline (Initial Shipment)
Sloth | Date of Death | Primary Diagnostic Consideration |
Selma | 08/16/2025 | Confirmed Gammaherpesviral Pneumonia |
Leif | 08/18/2025 | Suspected Gammaherpesvirus; gastric dilation |
Flash | 08/26/2025 | Clinical decline/autolysis (not submitted) |
Slakoth | 08/29/2025 | Suspected Gammaherpesvirus; peritonitis |
Sid | 09/09/2025 | Myelomalacia; lymphangiectasia; gastric dilation |
Belt | 09/11/2025 | Myelomalacia; lymphocytic encephalitis |
Poncho | 09/27/2025 | Clinical decline following coughing/lethargy |
Baloo | 11/02/2025 | Nephritis; esophagitis; bone marrow atrophy |
PCR testing performed by the University of Florida (UF) Zoo Med Diagnostic Lab identified a novel two-fingered sloth gammaherpesvirus as the presumptive underlying cause for the high mortality rate.
SUMMARY OF FINDINGS
Necropsies conducted by the Bronson Animal Disease Diagnostic Laboratory (BADDL) revealed consistent multi-organ involvement across numerous submissions.
Respiratory: Interstitial and Bronchopneumonia
Most sloths exhibited lungs with multifocal, dark-red, consolidated regions.
- Selma (K2511058): Severe, bilateral interstitial pneumonia with fibrin and edema.
- Gator, Tugboat, Diablo (K2600020): All showed pneumonia; two were confirmed positive for herpesvirus
Hematopoietic: Bone Marrow and Splenic Depletion
A recurring finding was moderate-to-severe hematopoietic depletion and serous fat atrophy within the bone marrow. This indicates a “negative energy balance with declining body condition and/or viral injury to proliferating hematopoietic cells.
- Gastrointestinal: Gastric Dilation and Hyperkeratosis
- Gastric Dilation: Many sloths (Leif, Slakoth, Sid, Belt, Sonic) presented with stomachs occupying 50-60% of the abdominal cavity.
- Hyperkeratosis: “Severe cobble stone squamoid proliferation” of the stomach mucosa was noted in several cases (Sonic, Snuggles).
Neurological: Spinal Cord Hemorrhage and Myelomalacia
- Spinal Hemorrhage: Extensive extradural hemorrhage was found in several sloths (Banksy, Picasso, Siesta, Diablo).
- Myelomalacia/Spongiosis: Severe vacuolization of the white matter (myelomalacia) was confirmed in Sid and Belt.
Nutritional and Mineral Deficiencies
Testing conducted at Michigan State University (MSU) revealed critical nutritional deficiencies that may have predisposed the sloths to the observed pathologies.
Vitamin A Deficiency and Hyperkeratosis
Testing for sloths Snuggles, Chili, and Sonic indicated a likely Vitamin A deficiency.
Copper Deficiency and Myelomalacia
Liver mineral analysis for sloths Gator, Tugboat, and Diablo showed copper levels (8.97 µg/g) significantly below the adequate range for domestic species. Pathologists noted that spinal cord myelomalacia in other species (known as “enzootic ataxia”) is specifically associated with copper deficiency.
Secondary Comorbidities – Bacterial and Fungal Pathogens
- Aliarcobacter butzleri: Recovered from the stomach of Nell; known to cause colitis in primates.
- Opportunistic Colonization: Aerobic cultures frequently identified Escherichia coli, Aeromonas hydrophila, Streptococcus gallolyticus, and Pseudomonas aeruginosa.
- Candidiasis: Baloo and Belt showed evidence of Candida overgrowth, potentially exacerbated by antibiotic administration (Baytril).
Parasitic Infestations – Heavy parasitic loads were common despite deworming protocols (Panacur, Ponazuril):
- Strongyles: “Too numerous to count” in Leif; identified in Belt.
- Coccidia: Eimeria identified in Baloo, Mossy, and Sonic.
- Sarcocystis: Identified in the tongue of Nell and the skeletal muscle of Diablo.
- Ticks: An engorged tick was removed from Slakoth, though the PCR panel for tick-borne diseases was negative.
Facility Protocols and Husbandry
The Program of Veterinary Care (USDA) for the facility, established August 15, 2025, by Dr. Jennifer Lane, outlines the following standards:
- Quarantine: Minimum 30-day period for new arrivals.
- Environmental Control: Temperature maintained at (23.89° – 29.44°) 75-85°F; humidity at 60-80%.
- Diet: Rotating diet of produce (squash, kale, sweet potato), specialized browse (papaya leaves, cecropia, mulberry), and Mazuri leaf eater biscuits.
- Monitoring: Visual exams twice daily; non-invasive temperature checks; weekly weights using low-stress “basket” techniques.
Stress, Housing, Handling, and Diet Related Sickness
Gastric Dilation in sloths, often progressing to volvulus, is a life-threatening condition reported in captive two-fingered sloths. There are multifactorial issues commonly known as dysbiosis. Predisposing factors involve improper diets, low temperature, and husbandry practices. Symptoms can vary from subtle to acute (Oliveira et al., 2024a).
Hyperkeratotic gastritis, particularly identified in recent studies of two-fingered sloths, is a rare, chronic condition characterized by thickening of the stomach lining (hyperkeratinization of the squamous epithelium) and excessive proliferation of the gastric mucosa. It involves chronic inflammation, likely linked to immune-mediated responses to captivity stress or dietary issues. This is a classic of chronic stress, poor nutrition, and health. Peritonitis, also mentioned in the report, occurs as a secondary complication of another abdominal disease rather than as a primary process (De et al., 1999a; de Stefani Munaó Diniz & Oliveira, 1999). Specific reference ranges for Vitamin A levels in sloths have not been established in the available scientific literature. However, clinical reports indicate that symptoms such as hyperkeratosis are more frequently associated with stress and inadequate nutrition, as documented in medical case studies of captive individuals (Almeida V.C. et al., 2023; De et al., 1999b; Digeronimo et al., 2023; Oliveira et al., 2024b). Stress, habitat changes, captive duration, and long-hour transportation can cause all the gastrointestinal symptoms previously reported in the pathological report (Thacher et al., n.d.).
It is also important to mention that one individual presented symptoms of gastrointestinal disease prior to being transported and was still transported from one country to another, resulting in easier sickness transmission and a lack of animal welfare. The reports openly mention that individuals experienced stress during transportation.
Myelomalacia is a recognized neuropathology in mammals; the most reported mechanisms are severe spinal trauma, hypoxia-ischemia, and embolic disease (Lin et al., 2023). In sloths, this kind of damage can be associated with rough handling, falls, or thoracolumbar injuries (Esplin, DlXON, M. WOODBURY, 1946). Due to their low metabolic rate and unique thoracic biomechanics, even partial cord injury may have a disproportionate impact on mobility and ventilation (Vigil-Esquivel et al., 2021), which could possibly explain many of the other findings in the pathological report.
Spinal cord hemorrhage in sloths is rare but serious condition often linked to trauma, infection or falls, rescue centers have reports spinal injuries after falling, leading to hemorrhage paralysis or euthanasia if untreated (Almeida V.C. et al., 2023; Burns et al., 2012; Chinchilla-Barboza et al., 2021). Sloths’ unique vertebral anatomy, with fewer cervical vertebrae and a specialized structure, may increase vulnerability to such damage (Chinchilla-Barboza et al., 2021). Spine damage often requires supportive care, and severe hemorrhage leads to poor prognosis even in humans (Almeida V.C. et al., 2023; Burns et al., 2012). In particular, there has been one report of Streptococcus agalactiae causing septicemia leading to hemorrhage in the brain (Yoshida et al., 2022). Necropsy examinations confirmed the presence of Streptococcus gallolyticus. While this bacterium is generally recognized as a gut commensal in mammals, particularly ruminants, it has been documented to cause septicemia under stress conditions (Cruz et al., n.d.). The clinical manifestations are most frequently associated with endocarditis (Gray et al., 2025). Furthermore, reports indicate that certain subspecies may be implicated in sepsis and meningitis in neonatal care settings (Hurrell & Ayyash, 2021).
The necropsy report noted spinal injuries, with conspecific aggression or trauma initially considered as possible causes. However, the frequency of such injuries appears unusually high, so they try to dismiss it as a cause. Nevertheless, behavioral records indicate that males were housed together along with several other sloths. It is well established that male sloths are highly territorial and prone to aggressive interactions, whereas females generally tolerate only brief contact during breeding (Care, 2021; Cliffe, n.d.; Dial, 2019; Dial et al., 2026; The Educated ZooKeeper LLC, 2021). Housing wild-caught sloths, each with established territories prior to capture, in shared enclosures inevitably increases the risk of aggression, which can result in severe trauma and significant injuries (Cliffe, n.d.; Dial, 2019; Dial et al., 2026). The practice of housing sloths together reflects a lack of consideration for the species’ natural history and territorial behavior. Housing several individuals together is recommended only if they have sufficient space (Dial, 2019; Dial et al., 2026).
Pulmonary congestion in sloths can occur secondary to another underlying disease, such as pneumonia, sepsis, or trauma (Vigil-Esquivel et al., 2021). Bronchopneumonia in sloths is a significant respiratory condition linked to bacterial infections; the literature specifically has reported Bordetella bronchiseptica, exacerbated by stress, poor housing, or captivity acclimation (Hammond et al., 2009). Although the agent is not mentioned in the reports, the specific bacterial agents identified in the lungs included Citrobacter, Enterococcus gallinarum, Escherichia coli, and Acinetobacter. Pathologists also frequently noted unidentified rods and coccobacilli in lung tissues, but none were confirmed to be Bordetella. Studies on two and three-fingered sloths report E. coli and Citrobacter isolates (Fernandes et al., 2022). E. gallinarum is not explicitly documented in sloths and may relate to captive exposure rather than the natural microbiome (Chaverri et al., 2025). Acinetobacter lacks direct sloth reports (Chaverri et al., 2025) but causes nosocomial respiratory infections in exotic mammals, aligning with the sloth bronchopneumonia reported (Baranzelli et al., 2013; van der Kolk et al., 2019).
On secondary bacterial pathogenic infection, Aliarcobacter butzleri, Aeromonas hydrophila and Pseudomonas aeruginosa there is no direct link to sloths, but its presence in rehab settings via contaminated water or food can be a proven risk of dysbiosis in stressed individuals (Fernandes et al., 2022; Miller et al., 2024). Quarantine and water hygiene are key preventives (Dial et al., 2026).
Finally, the report identified nutritional and mineral deficiencies, specifically involving Vitamin A and copper. However, no published reference ranges exist for sloths, making it difficult to assess the clinical relevance of these findings. Given the multiple stressors and symptoms documented throughout these reports, these deficiencies may represent incidental observations rather than primary pathological factors.
Reports of Viral Findings
As mentioned before the reports evaluates the thesis that the primary driver of this mortality event is the recrudescence of a novel viral infection.
The identified viruses are:
- Gammaherpesvirus
- Changuinola virus
More viruses are mentioned but not properly identified yet.
Reported in sloths Gammaherpesvirus causes oral vesicular lesions and oral candidiasis the lesions that are not fully comunicated in the necropsy reports which could make the viral incidental (Sacristán et al., 2026). The sloth in the literature show persistent oral lesions, but routine bloodwork and organ-function tests were unremarkable, suggesting that gammaherpesvirus may act as a co-factor or opportunistic pathogen rather than a consistent severe primary disease. The reports do not establish a direct association between the observed candidiasis and the presence of gammaherpesvirus, despite clinical evidence documenting similar pathogenicity in comparable cases. This lack of correlation warrants further consideration, given that candidiasis has been clinically reported in conjunction with gammaherpesvirus infections under analogous conditions (Benarrós & Salvarani, 2024; Sacristán et al., 2026). Because they are often latent and can reactivate under stress or immunosuppression, similar infections in wild or rehabbed sloths might be under-recognized, especially when lesions are mild or intermittent (Benarrós & Salvarani, 2024; Sacristán et al., 2026).
Changuinola is an arthropod-borne virus, it circulates in Central and South America with reports from regions such as Panama, Colombia and Brazil. In humans it is commonly known as Changuinola fever, typically presented as self-limited febrile illness. The most common reservoir and natural host is wild rodents and small mammals. In this context, arboreal mammals like sloths may act as incidental or spill over hosts, given their overlapping habitat with sand-fly vectors and other infected mammals in tropical forest ecosystems (Silva et al., 2013, 2014). The transportation of unscreened wild caught viremic individual to a nation where the disease has not been reported is a One Health concern that may aid in the disease’s spread. Sloths can develop viremia and virus circulation, but these individuals have been reported as clinically healthy or only subclinical affected suggesting that Changuinola virus disease is usually mild or inapparent in this taxonomic group. However, in a stress or immunocrompromised individual (malnourished), the same virus could potentially contribute to systemic illness, though severe cases have not been well documented specifically in sloths (Corrales et al., 2025).
Viruses have been detected in wild sloths, yet their role as disease-causing agents remains undemonstrated. Current evidence suggests that viral presence is a natural component of sloths’ ecology, forming part of their microbiome and wildlife health dynamics(Corrales et al., 2025; de Oliveira Filho et al., 2020).
There is significant importance in reporting the presence of viruses in sloths in these pathology reports which could potentially guide future discoveries of viral presence in sloths. However, it is incorrect to only attribute any coincidental lesion, sickness or change in health to only a viral infection. These animals had to deal with a variety of stressors. Such as wild capture and captivity, nutritional changes, and environmental pressure changes brought on by moving from a tropical habitat to a captive environment in a different nation in a short period of time, all these factors can all have a detrimental impact on their health and confuse interpretations of viral impact to a systemic failure that addresses a number of other issues.
Conclusion
The mass mortality event among two-toed sloths transported from Peru to Florida in 2025–2026 reflects a multifactorial health crisis driven by the interaction of infectious, nutritional, environmental, and husbandry-related stressors. Necropsy and diagnostic investigations consistently identified severe systemic pathology, including interstitial pneumonia, gastric dilation, hyperkeratosis, myelomalacia, and hematopoietic depletion. A novel gammaherpesvirus was detected and considered a primary contributor; however, its role appears opportunistic, exacerbated by capture stress, cold exposure during transport, and compromised immunity. Secondary bacterial, fungal, and parasitic infections further complicated clinical outcomes, while nutritional deficiencies in copper and Vitamin A likely predisposed individuals to neurological and gastrointestinal disease, though their relevance remains uncertain due to the absence of species-specific reference ranges.
The evidence demonstrates that inadequate husbandry practices, including housing territorial males together, transporting clinically compromised individuals, and failing to mitigate environmental stressors, significantly increased morbidity and mortality. These findings underscore the importance of aligning captive management protocols with the species’ natural history, implementing rigorous quarantine and screening procedures, and ensuring nutritional adequacy. Viral presence, while notable, should not be interpreted in isolation; rather, the cumulative impact of stress, poor housing, and nutritional imbalance must be recognized as central to the systemic failure observed.
This event highlights critical gaps in knowledge regarding sloth physiology, nutritional requirements, and disease susceptibility where standard of care for captive sloths wasn’t met. It emphasizes the urgent need for improved welfare standards, stricter transport regulations, and enhanced surveillance of emerging pathogens in wildlife trade. Addressing these deficiencies is essential to prevent recurrence, safeguard animal welfare, and mitigate broader One Health risks associated with the movement of wild-caught species across international borders.
Dra. Ana Maria Villada
Analysis of Pathology Reports Made By:
Ana María Villada Rosales
- Head Veterinarian and Conservation Medicine Research Manager, The Sloth Institute Costa Rica
- PhD Student Conservation Medicine, Universidad Andrés Bello
- Master in Conservation Medicine, Murdoch University
- Representative NGO member of the Council of Scientific Authority in Costa Rica (CRACCITES)
- Active member of the National Council of Wildlife Medicine in Costa Rica (CONMVEFAS)
- Latin American member of Wildlife Disease Association (WDA)